Human Organoids #
Organoids are three-dimensional, self-organizing cultures derived from stem cells that recapitulate the structure and function of human organs. This section showcases how organoids are transforming disease modeling, drug screening, and gene therapy development, enabling precision medicine approaches for conditions like cystic fibrosis, Duchenne muscular dystrophy, and cancer.
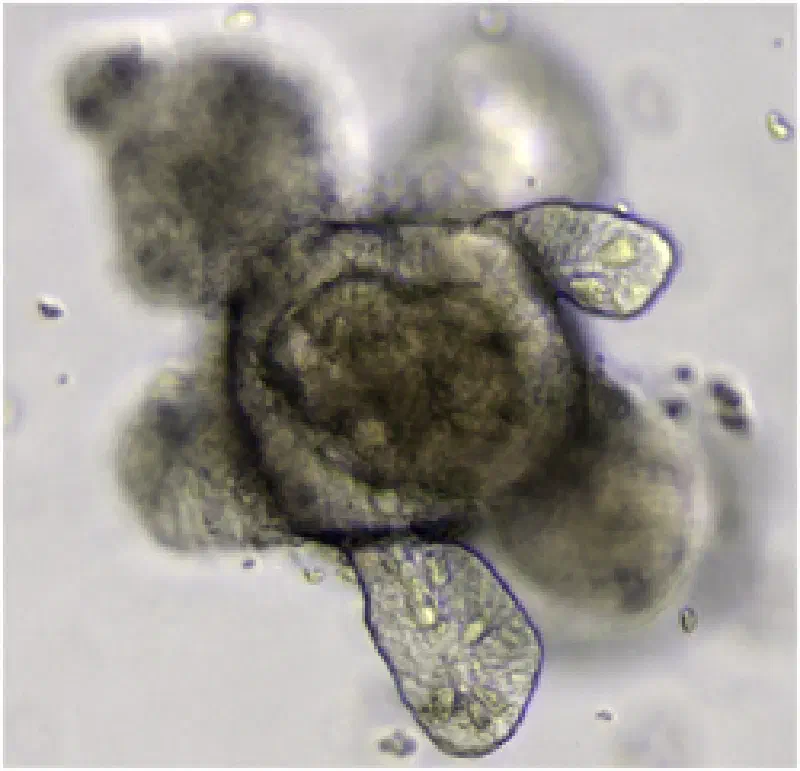
from Lgr5+ stem cells.
Credit: Meritxell Huch, CC BY 4.0
FIS Assay for Cystic Fibrosis #
Patient-derived intestinal organoids from Cystic Fibrosis patients were used in the Forskolin-induced Swelling (FIS) assay to test CFTR-modulator drugs. The assay accurately predicted clinical trial responses for individual patients, including those with rare genotypes. This has enabled tailored therapeutic strategies, significantly increasing life expectancy for Cystic Fibrosis patients.
Towards diagnostic and personalized models using organoids
NAMs: an exciting era for drug discovery
Patient-Derived Organoids for Gene Therapy in DMD #
A breakthrough workflow successfully converted cryopreserved blood cells into induced pluripotent stem cells and then into cardiac organoids, correcting unique splicing defects in Duchenne Muscular Dystrophy patients. Custom ASOs restored dystrophin expression and improved calcium transients in these cardiac organoids, validating the therapeutic approach. This provided a scalable, cost-effective alternative to animal models for developing personalized gene therapies.
Patient-Derived Organoids for Gene Therapy Development
Patient-Derived Organoids for Metastatic Colorectal Cancer #
The OPTIC trial validated the predictive power of patient-derived organoids (PDOs) for metastatic colorectal cancer, showing that organoid response correlated with radiological tumor response and clinical survival. The trial demonstrated 83.3% accuracy in predicting patient survival and tumor response. This has enabled early identification of ineffective therapies, minimizing patient exposure to toxicity and optimizing treatment selection.
Patient-Derived Organoids Predict Treatment Response in Metastatic Colorectal Cancer
Organoid Immune Co-Culture Models for Cancer Vaccines #
Tumor organoids co-cultured with autologous peripheral blood lymphocytes simulated the tumor immune microenvironment to assess individual responses to checkpoint inhibitors and CAR-T cell therapies. This method identified tumor-specific antigens with high immunogenicity, enabling the design of personalized cancer vaccines. The discovery has revolutionized immunotherapy development by capturing spatial organization and immune dynamics.
From petri dish to patient care: organoids bring personalised cancer therapy closer
Kidney Assembloids for Polycystic Kidney Disease #
Researchers generated the most complex kidney structures to date—assembloids combining filtering nephrons with urine-concentrating collecting ducts. These assembloids recapitulated key features of Polycystic Kidney Disease, including inflammation and fibrosis, which were previously irreproducible in animal models. This work opened new avenues for studying chronic kidney disease and predicting drug-induced nephrotoxicity.
Researchers develop most advanced kidney organoid yet for disease modeling and drug discovery
Miller-Dieker Syndrome Root Cause #
Human brain organoids derived from patient cells identified the root cause of Miller-Dieker Syndrome as early neural stem cell death and severe division defects in “outer radial glia.” Time-lapse imaging showed that these specific glia cells—which are entirely absent in mouse models—failed to divide properly. This solves a long-standing mystery in neurodevelopmental disease and proves that patient-derived organoids can bridge the gap between animal models and human pathophysiology.
An Organoid-Based Model of Cortical Development Identifies Non-Cell-Autonomous Defects in Wnt Signaling Contributing to Miller-Dieker Syndrome
Cerebral organoids expressing mutant actin genes reveal cellular mechanism underlying microcephaly
IGF-1 Dependency in Lung Cancer Subtypes #
A library of 40 small cell lung cancer (SCLC) organoid lines revealed that non-neuroendocrine subtypes depend on the IGF-1 signaling axis for growth. Genetic ablation in human alveolar organoids replicated this dependency, and IGF1R inhibitors suppressed growth in patient-derived models. This identifies IGF1R inhibition as a promising new therapeutic strategy for a treatment-resistant patient population.
An organoid library unveils subtype-specific IGF-1 dependency via a YAP–AP1 axis in human small cell lung cancer
Intestinal Organoids Reveal Stem Cell Biology #
The first human intestinal organoids developed from adult stem cells enabled the study of gut disease, cancer, and drug responses in human tissue. This success was extended to liver, kidney, brain, and retinal organoids worldwide, validating the platform across multiple organ types. This technology serves as the foundation for modern human organoid research.
Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche
Brain Organoids Model Microcephaly Caused by Zika Virus #
Human brain organoids demonstrated Zika virus infection of neural progenitor cells, modeling microcephaly-like features in human tissue. The organoids captured human-specific features of microcephaly that mouse models were unable to fully recapitulate. This provided critical, human-specific insights into Zika virus pathology.
Brain-Region-Specific Organoids Using Mini-bioreactors for Modeling ZIKV Exposure
Zika virus tested in human brain organoids
Understanding Brain Development and Disease #
Brain organoids, three-dimensional, laboratory-grown models derived from induced pluripotent stem cells that replicate the complex, species-specific developmental programs of the human brain, have revolutionized neuroscience by allowing researchers to study the unique expansion of the human cortex. Organoids serve as critical tools for modeling neurodevelopmental disorders such as autism and microcephaly, testing treatments, and exploring evolutionary differences through the integration of ancient hominin genes. Regional organoids are fused into “assembloids” to map functional neural circuits, including motor-muscle connections and pain-processing pathways. Despite their transformative potential, organoids currently lack the full complexity of a natural brain and face long-term sustainability challenges, which, alongside improving technology, necessitates ongoing ethical scrutiny regarding the future possibility of emergent sentience.
Mini models of the human brain are revealing how this complex organ takes shape