Regulatory & Industry Adoption of NAMs #
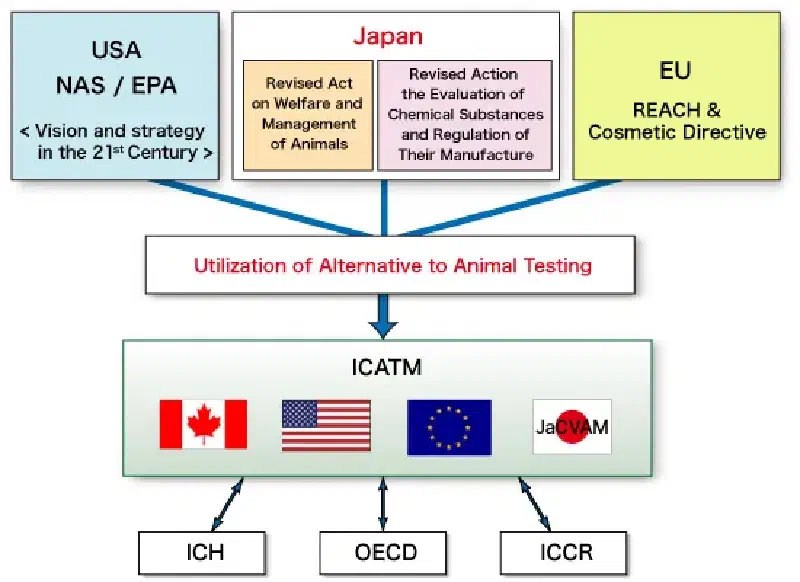
Alternative Test Methods (ICATM)
Credit: National Institute of Health Sciences
Regulatory agencies and pharmaceutical companies are increasingly embracing NAMs to streamline drug development, enhance predictivity, and align with ethical and scientific advancements. This section examines key milestones—such as the FDA Modernization Act 2.0 and OECD standards—that are paving the way for the global adoption of human-relevant, animal-free methodologies in biomedical research.
FDA Modernization Act 2.0 #
The FDA Modernization Act 2.0 (2022) removed the federal mandate for animal testing in new drug applications. This was a significant move by the FDA toward utilizing human cells and organoids in preclinical safety assessments. The act explicitly encouraged the use of NAMs to provide more accurate and relevant data for human health.
FDA Modernization Act 2.0: transitioning beyond animal models with human cells, organoids, and AI/ML-based approaches
How new approach methodologies are reshaping drug discovery
Organ-on-a-chip meets artificial intelligence in drug evaluation
FDA CDER NAM Validation Guidance #
The FDA’s Center for Drug Evaluation and Research (CDER) released draft guidance establishing a validation framework for NAM-derived data. These guidelines are based on scientific confidence and “fit-for-purpose” utility, marking a definitive shift away from the animal model. This provided a clear regulatory pathway for integrating NAMs into Investigational New Drug applications.
FDA Releases Draft Guidance on Alternatives to Animal Testing in Drug Development
New Approach Methodologies (NAMs) in Drug Development
OECD International Standards #
The OECD Guidance Document 34 established international standards for the validation and acceptance of alternative test methods. This facilitated global harmonization and the widespread adoption of NAMs in various regulatory frameworks. These standards ensure that non-animal data is accepted consistently across member countries.
List of Alternative Test Methods and Strategies (or New Approach Methodologies NAMs)
OECD Series On Testing And Assessment Number 34
Tebentafusp (Kimmtrak) Regulatory Approval #
Tebentafusp (Kimmtrak) became the first immunotherapy to reach regulatory approval without in vivo animal pharmacodynamic data. Because the drug lacked activity in any animal species, the sponsors relied entirely on human-centric NAMs to justify safety and efficacy. This establishes a major regulatory milestone, proving that human-relevant data can fully replace animal testing for specific first-in-class therapeutics.
Immunocore announces FDA approval of KIMMTRAK® (tebentafusp-tebn) for the treatment of unresectable or metastatic uveal melanoma
Summary Basis of Decision for Kimmtrak in Canada
USP Chapter <86> Recombinant Reagents #
The U.S. Pharmacopeia implemented Chapter <86> in May 2025, endorsing the use of non-animal-derived reagents for bacterial endotoxin testing. This change allows for the use of recombinant Factor C, utilizing gene sequences from horseshoe crabs instead of their blood. This modernization could save up to 90,000 animals per year while increasing batch consistency in pharmaceutical manufacturing.
<86> Bacterial Endotoxins Test Using Recombinant Reagents